3D Printed – which thickness is best for my patient?
One of the common questions we get from clients who are prescribing 3D printed orthotics for the first time is “what thickness should I use?” Most podiatrists are familiar and comfortable with the material properties of polypropylene, subortholen, carbon fibre and EVA. But prescribing a new material can be daunting!
QOL prints in a material called Polyamide 11 (PA 11) or Nylon 11 (then branded internally as EnviroPoly). PA 11 is a polyamide, bioplastic and a member of the nylon family of polymers produced by the polymerization of 11-aminoundecanoic acid. It is produced from castor beans by Arkema, a global specialty chemicals and advanced materials company. PA 11 was around well before 3D printing – the first patents were filed in 1947.
Fundamentally, PA 11 behaves in a similar fashion to polypropylene. We’ve created this handy guide to help you navigate which thickness is best with an orthotic without any extrinsic posting:

So if you would have previously prescribed a 3mm polypropylene device, the equivalent in PA 11 would be about 2.7-2.8mm. Or if you would have previously prescribed the polyflex device (2mm polypropylene with a 3mm EVA ILA filler), the equivalent thickness in PA 11 would be about 2.3-2.4mm.
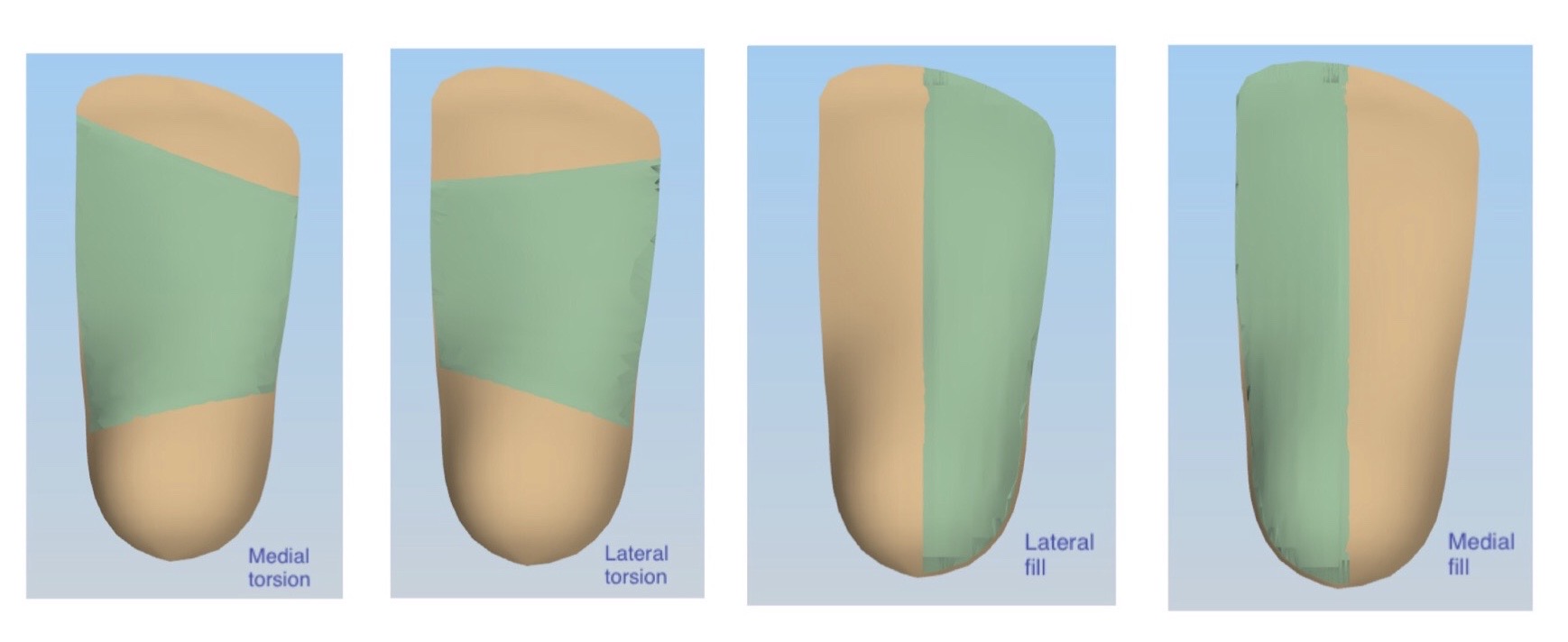
One of the main advantages in prescribing PA 11 devices though, lie more in the fact that we’re utilizing additive manufacturing production – meaning we’re literally starting with nothing and adding layer by layer of powder to build the orthotic from the ground up. This means we’re not constrained to having the same thickness of material throughout the orthotic.
The design options for 3D printed orthotics are near endless. If you want to have a shell which is 2.7mm thick on the medial side and 4.2mm thick on the lateral side to control lateral instability – that’s no problems. Or if you want to taper the distal medial edge from 3.2mm to 1.5mm to improve first ray function – that’s no problem. The ability to vary thickness throughout the device is near limitless.
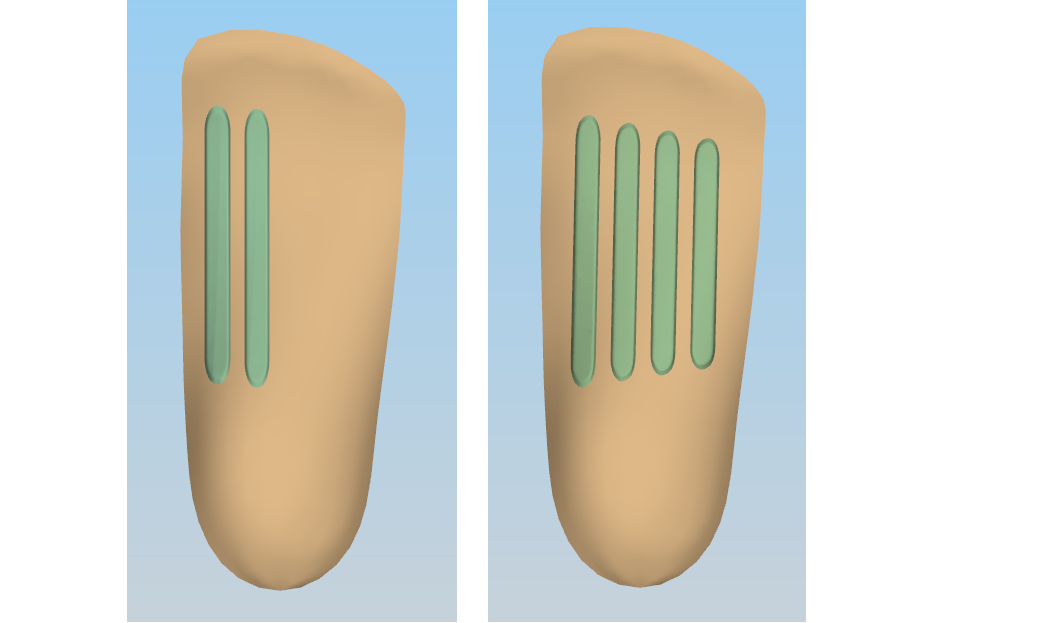
Similarly, the ability to add stiffening components to the plantar surface of the device are an exciting breakthrough in prescribing custom orthotics. We’ve trailed a range of different stiffening techniques which allows the treating podiatrist to keep the shell thin and reduce the bulk for shoe fit, but reinforce and stiffen the device in the critical areas for the patient.
If you would like to discuss integrating 3D printing into your practice, contact Kent by email at [email protected], or make a booking here.